Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (24): 3912-3918.doi: 10.3969/j.issn.2095-4344.2014.24.023
Previous Articles Next Articles
Progress of proteomics technology and its application in osteoarthritis
Pan Ding, Lei Guang-hua
- Department of Orthopedics, Xiangya Hospital of Central South University, Changsha 410008, Hunan Province, China
-
Revised:2014-05-23Online:2014-06-11Published:2014-06-11 -
Contact:Lei Guang-hua, M.D., Professor, Department of Orthopedics, Xiangya Hospital of Central South University, Changsha 410008, Hunan Province, China -
About author:Pan Ding, M.D., Physician, Department of Orthopedics, Xiangya Hospital of Central South University, Changsha 410008, Hunan Province, China -
Supported by:the National Natural Science Foundation of China, No. 30300396
CLC Number:
Cite this article
Pan Ding, Lei Guang-hua . Progress of proteomics technology and its application in osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(24): 3912-3918.
share this article
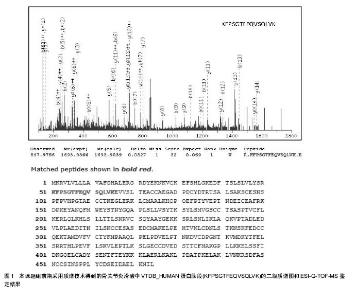
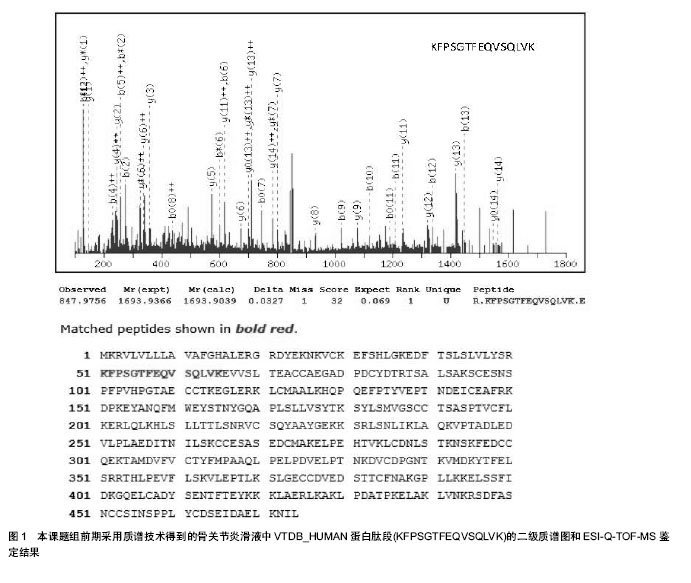
2.1 蛋白质组学概述 蛋白质组是指由一个基因组,或一个细胞、组织表达的所有蛋白质[19]。蛋白质组的概念与基因组的概念有许多差别,它随着组织结构、甚至环境状态的不同而改变。一个基因在转录后可以多种mRNA形式进行剪接,并且同一个蛋白质也可能以许多形式进行翻译后的修饰。因此一个蛋白质组并不是一个基因组的直接产物,蛋白质组中蛋白质的数目可以超过基因组的数目[20]。蛋白质组学研究集中于动态地描述基因的各种调节,对基因的表达在蛋白质水平上进行定量地测定,鉴定药物和疾病对人体组织的影响,以及尝试解释基因表达调控的机 制[21-22]。蛋白质组学研究是基因产物图谱和蛋白质(多肽)图谱技术的一种延伸,基因产物图谱依靠多种物理化学方式分离后的分析,如质谱技术和氨基酸组分分析等[23];而蛋白质(多肽)图谱则依靠双向凝胶电泳技术和进一步的图象分析。 国际上蛋白质组学研究已经取得了一定的进展,不论是在基础理论方面还是技术方法上,都在不断完善和进步。目前已经建立了多种蛋白质组学数据库。1996年,澳大利亚建立了世界上第一个蛋白质组研究中心:Australia Proteome Analysis Facility (APAF) [24]。2001年4月,在美国成立了国际人类蛋白质组研究组织(Human Proteome Organization, HUPO) [25],随后欧洲、亚太地区都相继成立了区域性蛋白质组研究组织,尝试通过合作的方式,融合各方面的力量,完成人类蛋白质组计划(Human Proteome Project) [26]。 2.2 蛋白质组学研究内容 2.2.1 蛋白质鉴定 利用1D电泳和2D电泳并结合免疫组织化学和Western blot等技术,或者利用免疫共沉淀、蛋白质芯片及抗体芯片等技术对组织中的蛋白质进行筛查和鉴定[27]。 2.2.2 蛋白质翻译后修饰 蛋白质在经过mRNA表达后,还需经历复杂的翻译后修饰如糖基化,磷酸化和酶原激活等。翻译后修饰是调节蛋白质功能的一种重要途径,因此对蛋白质翻译后修饰的研究对阐明蛋白质的功能具有重要作用[28]。 2.2.3 蛋白质功能分析 主要包扩酶活性的分析和鉴定对应酶的底物,以及细胞因子功能网络或信号转导通路的分析。利用的技术主要包括基因敲除技术和反义表达技术。此外研究表达蛋白在细胞中的定位在一定程度上也有益于蛋白质的功能分析,比如Clontech公司研发的荧光蛋白表达系统就是研究细胞内蛋白质定位的一种工具[29]。 2.2.4 靶蛋白的寻找 蛋白质是很多药物的单一或复合组分,而很多药物的受体或靶分子也是蛋白质。除了直接作用于靶蛋白外,也可以通过对靶蛋白的研究来设计出干预蛋白质-蛋白质相互作用的药物分子[30]。 了解和探索人类不同生长、发育期和不同生理、病理条件下及不同细胞类型的基因表达特点,对于基础和临床医学病理机制研究具有特别重要的意义。通过相关蛋白质组学研究可以找到直接与特定的与生理病理状态相关的蛋白质分子,为进一步设计作用于靶蛋白的药物奠定基 础[31]。 2.3 蛋白质组学技术分类 蛋白质组学技术的发展已经成为现代生物技术快速发展的重要支撑,在近些年骨代谢和骨生物学研究方向取得了关键性的突破,尤其是在骨组织相关细胞的胞内信号传导方面取得了一定的进展[32]。 2.3.1 双向凝胶电泳 双向凝胶电泳的原理是第一向基于蛋白质的不同等电点采用等电聚焦分离,第二向则按不同的分子量采用SDS-PAGE分离,把复杂蛋白混合物中的总蛋白在二维平面上分离。由于双向电泳技术在蛋白质组与医学研究中所处的重要位置,它可用于蛋白质转录及转录后修饰研究、蛋白质组的比较和蛋白质间的相互作用、细胞分化凋亡研究、致病机制及耐药机制的研究、蛋白纯化等许多方面。近年来经过多方面改进已成为研究蛋白质组的最有使用价值的核心方法[33]。 2.3.2 等电聚焦 等电聚焦是一种利用有pH梯度的介质分离不同等电点蛋白质的电泳技术。等电聚焦凝胶电泳依据蛋白质分子的静电荷或等电点进行分离,蛋白质分子在含有载体两性电解质形成的一个连续而稳定的线性pH梯度中电泳。蛋白质分子在偏离其等电点的pH条件下带有电荷,因此可以在电场中移动;当蛋白质迁移至其等电点位置时,其静电荷数为零,在电场中不再移动,据此原理将蛋白质分离[34]。 2.3.3 生物质谱 生物质谱技术是蛋白质组学研究中最重要的鉴定技术,其基本原理是样品分子离子化后,根据不同离子之间的荷质比(M/E)的差异来分离并确定分子量。对于经过双向电泳分离的目标蛋白质用胰蛋白酶酶解(水解Lys或Arg的-C端形成的肽键)成肽段,对这些肽段用质谱进行鉴定与分析[35]。目前常用的质谱技术包括以下两种:基质辅助激光解吸电离-飞行时间质谱(MALDI-TOF- MS)和电喷雾质谱(ESI- MS)(图1)。"
| [1] 李宁华,张耀男,张毅,等.国内六大行政区域六城市中老年人群膝关节骨性关节炎患病危险因素比较[J].中国组织工程研究与临床康复,2007,11(39):7758-7760. [2] Muraki S, Oka H, Akune T, et al. Prevalence of radiographic knee osteoarthritis and its association with knee pain in the elderly of Japanese population-based cohorts: the ROAD study. Osteoarthritis and cartilage.2009;17(9):1137-1143. [3] 王伟,王坤正,党小谦,等.中老年人人群骨关节炎的流行病学研究[J]. 中国老年学杂志,2007,27(6):566-568. [4] Goldring MB, Goldring SR. Osteoarthritis. Journal of cellular physiology.2007;213(3):626-634. [5] Kapoor M,Martel-Pelletier J,Lajeunesse D,et al.Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nature reviews. Rheumatology. 2011;7(1): 33-42. [6] Buckland J. Osteoarthritis: Complement-mediated inflammation in OA progression. Nature reviews. Rheumatology.2012;8(1):2. [7] Benito MJ, Veale DJ, FitzGerald O, et al. Synovial tissue inflammation in early and late osteoarthritis. Ann Rheum Dis.2005;4(9):1263-1267. [8] Afonso V, Champy R, Mitrovic D, et al.Reactive oxygen species and superoxide dismutases: role in joint diseases. Joint Bone Spine.2007;74(4):324-329. [9] Sutton S, Clutterbuck A, Harris P, et al.The contribution of the synovium, synovial derived inflammatory cytokines and neuropeptides to the pathogenesis of osteoarthritis. Vet J. 2009;179(1):10-24. [10] Pelletier JP, Martel-Pelletier J, Abramson SB. Osteoarthritis, an inflammatory disease: potential implication for the selection of new therapeutic targets. Arthritis Rheum. 2001; 44(6):1237-47. [11] Bondeson J, Wainwright SD, Lauder S, et al.The role of synovial macrophages and macrophage-produced cytokines in driving aggrecanases, matrix metalloproteinases, and other destructive and inflammatory responses in osteoarthritis. Arthritis Res Ther.2006;8(6):R187 [12] Goldring MB, Otero M. Inflammation in osteoarthritis. Current opinion in rheumatology. 2011;23(5):471-478. [13] Fiorito S,Magrini L,Adrey J,et al.Inflammatory status and cartilage regenerative potential of synovial fibroblasts from patients with osteoarthritis and chondropathy. Rheumatology (Oxford).2005;44(2):164-171. [14] Pearle AD, Scanzello CR, George S, et al.Elevated high-sensitivity C-reactive protein levels are associated with local inflammatory findings in patients with osteoarthritis. Osteoarthritis Cartilage. 2007;15(5):516-23. [15] Koopman W.Arthritis and Allied Conditions, 14thed . A Textbook of Rheumatology, vol.2. Lippincott Williams and Wilkins, Philadelphia.2001:2167-2209. [16] Hasegawa M, Segawa T, Maeda M, et al.Thrombin-cleaved osteopontin levels in synovial fluid correlate with disease severity of knee osteoarthritis. J Rheumatol. 2011;38(1): 129-134. [17] Honsawek S, Tanavalee A, Sakdinakiattikoon M, et al. Correlation of plasma and synovial fluid osteopontin with disease severity in knee osteoarthritis. Clinical biochemistry. 2009;42(9):808-812. [18] Gao SG, Li KH, Zeng KB, et al. Elevated osteopontin level of synovial fluid and articular cartilage is associated with disease severity in knee osteoarthritis patients. Osteoarthritis Cartilage. 2010;18(1):82-87. [19] Wilkins, M. Proteomics data mining. Expert Rev Proteomics. 2009;6(6):599-603. [20] Osman KT, Edwards A. Structural genomics of human proteins. Methods Mol Biol. 2014;1140:27-34. [21] Benevento M, Di Palma S, Snijder J, et al. Adenovirus Composition, Proteolysis and Disassembly Studied by in-depth Qualitative and Quantitative Proteomics. J Biol Chem. 2014;289(16):11421-11430. . [22] Baker P R, Chalkley RJ. MS-Viewer: A Web Based Spectral Viewer for Proteomics Results. Mol Cell Proteomics. 2014; 13(5):1392-1396. [23] Dunn MJ. PROTEOMICS Reviews 2014. Proteomics. 2014; 14(4-5):333-337. [24] Heazlewood JL, Millar AH. AMPDB: the Arabidopsis Mitochondrial Protein Database. Nucleic Acids Res.2005, 33(Database issue), D605-610. [25] Brower V. Proteomics: biology in the post-genomic era. Companies all over the world rush to lead the way in the new post-genomics race. EMBO Rep.2001;2(7):558-560. [26] Kelleher NL. A cell-based approach to the human proteome project. J Am Soc Mass Spectrom.2012;23(10):1617-1624. [27] Noordin R, Othman N. Proteomics technology - a powerful tool for the biomedical scientists. Malays J Med Sci. 2013; 20(2):1-2. [28] Haft DH, Payne SH, Selengut JD. Archaeosortases and exosortases are widely distributed systems linking membrane transit with posttranslational modification. J Bacteriol. 2012; 194(1):36-48. [29] Cao W, Epstein C, Liu H, et al. Comparing gene discovery from Affymetrix GeneChip microarrays and Clontech PCR-select cDNA subtraction: a case study. BMC Genomics. 2004;5(1):26. [30] Scott DE, Ehebauer MT, Pukala T, et al. Using a fragment-based approach to target protein-protein interactions. Chembiochem.2013;14(3):332-342. [31] Qing LS, Tang N, Xue Y, et al. Identification of Enzyme Inhibitors Using Therapeutic Target Protein - Magnetic Nanoparticle Conjugates. Anal Methods. 2012;4(6): 1612-1615. [32] Lee JH, Cho JY. Proteomics approaches for the studies of bone metabolism. BMB Rep. 2014;47(3):141-148. [33] Vensel WH, Tanaka CK, Altenbach SB. Protein composition of wheat gluten polymer fractions determined by quantitative two-dimensional gel electrophoresis and tandem mass spectrometry. Proteome Sci.2014;12(1):8. [34] Zhu G, Sun L, Yang P.On-line amino acid-based capillary isoelectric focusing-ESI-MS/MS for protein digests analysis. Anal Chim Acta.2012;750:207-211. [35] Xu M, Yang L, Wang Q. Chemical interactions of mercury species and some transition and noble metals towards metallothionein (Zn7MT-2) evaluated using SEC/ICP-MS, RP-HPLC/ESI-MS and MALDI-TOF-MS. Metallomics. 2013;5(7):855-860. [36] Balazova T, Makovcova J, Sedo O, et al. The influence of culture conditions on the identification of Mycobacterium species by MALDI-TOF MS profiling. FEMS Microbiol Lett. 2014;353(1):77-84. [37] Khemchyan LL, Khokhlova EA, Seitkalieva MM.Efficient Sustainable Tool for Monitoring Chemical Reactions and Structure Determination in Ionic Liquids by ESI-MS. Chemistry Open.2013;2(5-6):208-214. [38] Suzuki T, Maeda T, Grant S, et al. Confirmation of Fructans biosynthesized in vitro from [1-13C]glucose in asparagus tissues using MALDI-TOF MS and ESI-MS. J Plant Physiol. 2013;170(8):715-722. [39] Gobezie R, Millett PJ, Sarracino DS, et al. Proteomics: applications to the study of rheumatoid arthritis and osteoarthritis. J Am Acad Orthop Surg.2006;14(6):325-332. [40] Coffman FD. Chitinase 3-Like-1 (CHI3L1): a putative disease marker at the interface of proteomics and glycomics. Crit Rev Clin Lab Sci.2008;45(6):531-562. [41] Vincourt JB, Vignaud JM, Lionneton F, et al. Increased expression of matrilin-3 not only in osteoarthritic articular cartilage but also in cartilage-forming tumors, and down-regulation of SOX9 via epidermal growth factor domain 1-dependent signaling. Arthritis Rheum.2008;58(9): 2798-2808. [42] Ruiz-Romero C, Calamia V, Mateos J, et al.Mitochondrial dysregulation of osteoarthritic human articular chondrocytes analyzed by proteomics: a decrease in mitochondrial superoxide dismutase points to a redox imbalance.Mol Cell Proteomics.2009;8(1):172-189. [43] Nemirovskiy O, Li WW, Szekely-Klepser G.Design and validation of an immunoaffinity LC-MS/MS assay for the quantification of a collagen type II neoepitope peptide in human urine: application as a biomarker of osteoarthritis. Methods Mol Biol.2010;641:253-270. [44] Fernandez-Costa C, Calamia V, Fernandez-Puente P.Sequential depletion of human serum for the search of osteoarthritis biomarkers. Proteome Sci.2012;10(1):55. [45] Ikeda D, Ageta H, Tsuchida K, et al.iTRAQ-based proteomics reveals novel biomarkers of osteoarthritis. Biomarkers. 2013; 18(7):565-572. [46] Balakrishnan L, Bhattacharjee M, Ahmad S, et al. Differential proteomic analysis of synovial fluid from rheumatoid arthritis and osteoarthritis patients. Clin Proteomics.2014;11(1):1. [47] Mobasheri A. Applications of proteomics to osteoarthritis, a musculoskeletal disease characterized by aging. Front Physiol. 2011;2:108. [48] Ali M, Manolios N.Proteomics in rheumatology: a new direction for old diseases.Semin Arthritis Rheum.2005:35(2): 67-76. [49] Aviner R, Geiger T, Elroy-Stein O.Genome-wide identification and quantification of protein synthesis in cultured cells and whole tissues by puromycin-associated nascent chain proteomics (PUNCH-P). Nat Protoc.2014;9(4):751-760. [50] O'Farrell PH.Two-Dimensional Gel Electrophoresis and the Beginning of Proteomics. Clin Chem. 2014 Feb 25. [Epub ahead of print] No abstract available. [51] Ghareeb HO, Radke W. Characterization of cellulose acetates according to DS and molar mass using two-dimensional chromatography.Carbohydr Polym.2013; 98(2):1430-1437. [52] Flaherty RJ, Huge BJ,Bruce SM,et al.Nicked-sleeve interface for two-dimensional capillary electrophoresis. Analyst. 2013;138(13):3621-3625. [53] Mellors JS, Black WA, Chambers AG, et al. Hybrid capillary/microfluidic system for comprehensive online liquid chromatography-capillary electrophoresis-electrospray ionization-mass spectrometry.Anal Chem.2013;85(8): 4100-4106. [54] Aerts JT, Louis KR, Crandall SR, et al.Patch Clamp Electrophysiology and Capillary Electrophoresis-Mass Spectrometry Metabolomics for Single Cell Characterization.Anal Chem. 86(6):3203-3208. [55] Shi CH, Schisler JC, Rubel CE, et al. Ataxia and hypogonadism caused by the loss of ubiquitin ligase activity of the U box protein CHIP. Hum Mol Genet.2014;23(4): 1013-1024. [56] Jungo F, Bougueleret L, Xenarios I, et al. The UniProtKB/ Swiss-Prot Tox-Prot program: A central hub of integrated venom protein data. Toxicon.2012;60(4):551-557. [57] Kind T, Liu KH, Lee do Y, et al. LipidBlast in silico tandem mass spectrometry database for lipid identification. Nat Methods. 2013;10(8):755-758. [58] Sheynkman GM, Shortreed MR, Frey BL, et al. Large-scale mass spectrometric detection of variant peptides resulting from nonsynonymous nucleotide differences. J Proteome Res.2014;13(1):228-240. [59] Woo S, Cha S W, Merrihew G, et al. Proteogenomic database construction driven from large scale RNA-seq data.J Proteome Res.2014;13(1):21-28. [60] Llorens F, Hummel M, Pastor X, et al. Multiple platform assessment of the EGF dependent transcriptome by microarray and deep tag sequencing analysis.BMC Genomics. 2011;12:326. |
| [1] | Peng Zhihao, Feng Zongquan, Zou Yonggen, Niu Guoqing, Wu Feng. Relationship of lower limb force line and the progression of lateral compartment arthritis after unicompartmental knee arthroplasty with mobile bearing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1368-1374. |
| [2] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [3] | Liu Xiangxiang, Huang Yunmei, Chen Wenlie, Lin Ruhui, Lu Xiaodong, Li Zuanfang, Xu Yaye, Huang Meiya, Li Xihai. Ultrastructural changes of the white zone cells of the meniscus in a rat model of early osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1237-1242. |
| [4] | Liu Xin, Yan Feihua, Hong Kunhao. Delaying cartilage degeneration by regulating the expression of aquaporins in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 668-673. |
| [5] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [6] | Cao Xuhan, Bai Zixing, Sun Chengyi, Yang Yanjun, Sun Weidong. Mechanism of “Ruxiang-Moyao” herbal pair in the treatment of knee osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 746-753. |
| [7] | Li Yonghua, Feng Qiang, Tan Renting, Huang Shifu, Qiu Jinlong, Yin Heng. Molecular mechanism of Eucommia ulmoides active ingredients treating synovitis of knee osteoarthritis: an analysis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 765-771. |
| [8] | Song Shan, Hu Fangyuan, Qiao Jun, Wang Jia, Zhang Shengxiao, Li Xiaofeng. An insight into biomarkers of osteoarthritis synovium based on bioinformatics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 785-790. |
| [9] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [10] | Zheng Li, Li Dadi, Hu Weifan, Tang Jinlong, Zhao Fengchao. Risk assessment of contralateral knee arthroplasty after unilateral total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 374-379. |
| [11] | Lü Jiaxing, Bai Leipeng, Yang Zhaoxin, Miao Yuesong, Jin Yu, Li Zhehong, Sun Guangpu, Xu Ying, Zhang Qingzhu. Evaluation of internal fixation with proximal femoral nail antirotation in elderly knee osteoarthritis patients with femoral intertrochanteric fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 391-396. |
| [12] | Luo Anyu, Liu Hanlin, Xie Xiaofei, Huang Chen. Effect of antioxidant mixture on structural degeneration of an osteoarthritis rat model [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3625-3629. |
| [13] | Gao Kun, Chen Dayu, Zhang Yong, Liu Weidong, Sun Shufen, Lai Wenqiang, Ma Dujun, Wu Yihong, Lin Zhanpeng, Jiang Yinglu, Yu Weiji. Achyranthes bidentata alcohol extract inhibits extracellular matrix degradation of the cartilage by regulating synovial fibroblast exosomes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3636-3640. |
| [14] | Liu Jinfu, Zeng Ping, Nong Jiao, Fan Siqi, Feng Chengqin, Huang Jiaxing. Integrative analysis of biomarkers and therapeutic targets in synovium of patients with osteoarthritis by multiple microarrays [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3690-3696. |
| [15] | Chen Feng, Zhang Xiaoyun, Chen Yueping, Liao Jianzhao, Li Jiajun, Song Shilei, Lai Yu. Molecular mechanism of anhydroicaritin in the treatment of osteoarthritis: an analysis based on network pharmacology and bioinformatics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3704-3710. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||